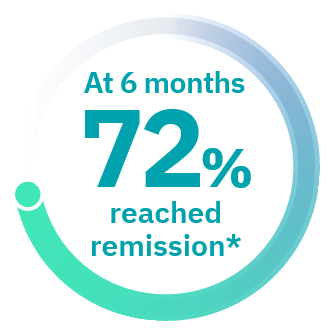
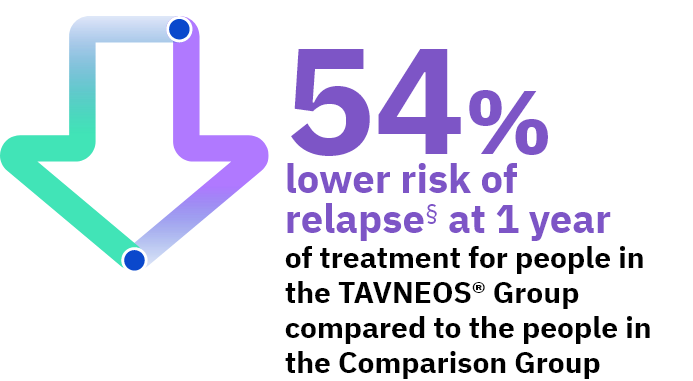
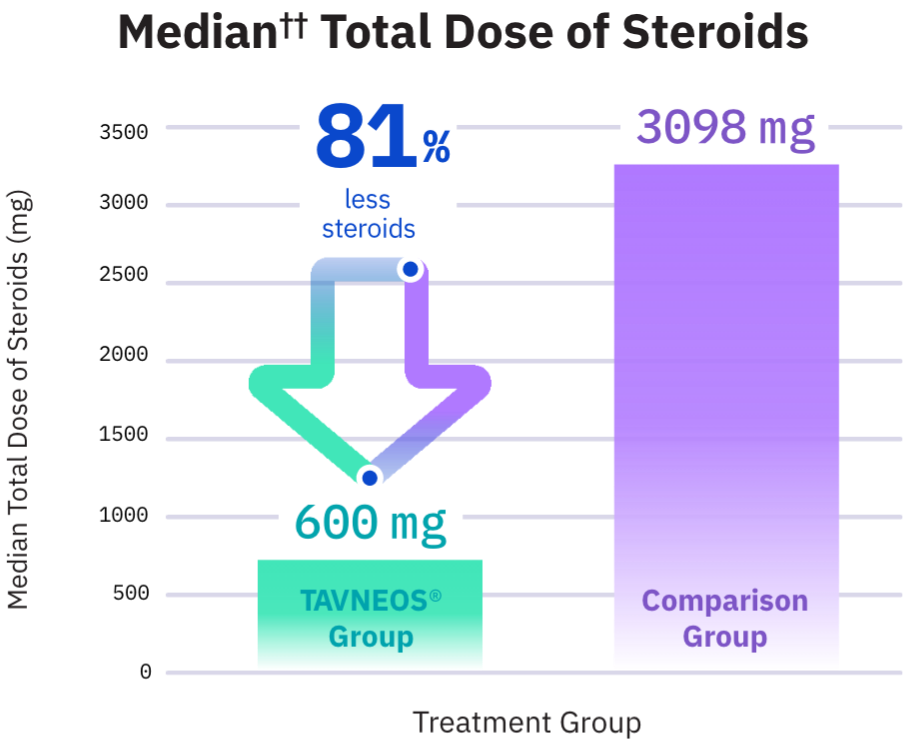
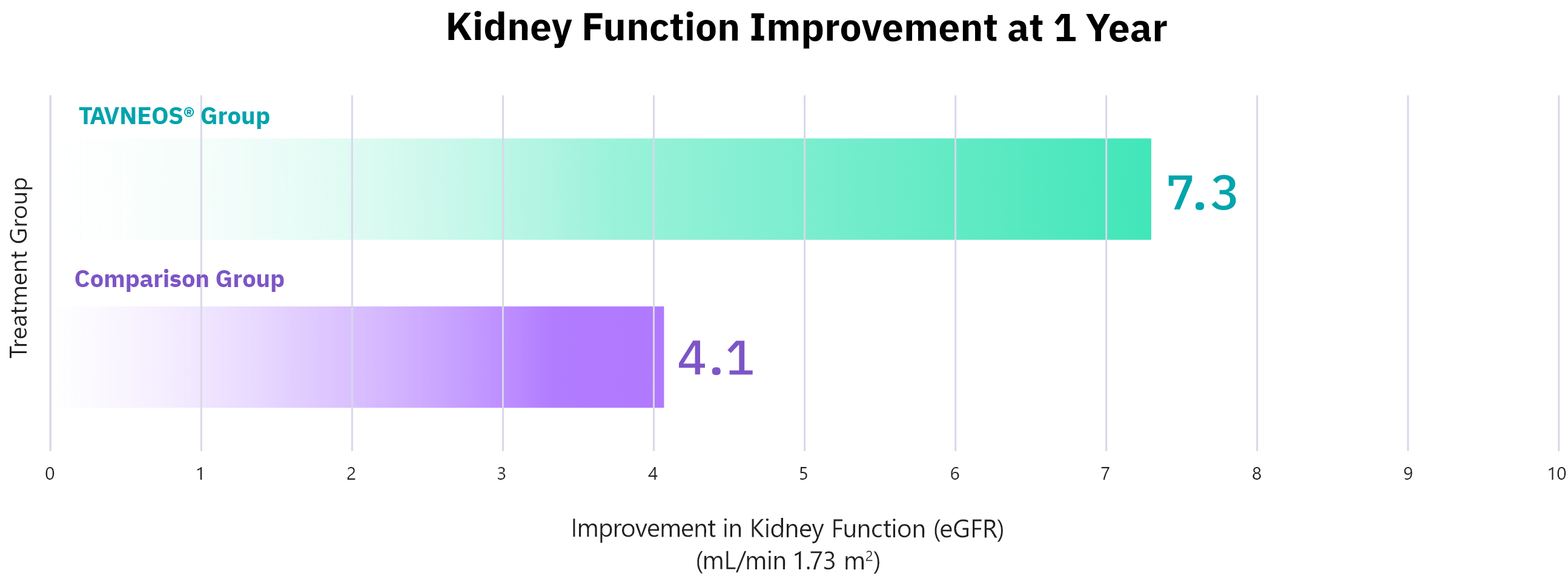
Approved Use: TAVNEOS® is a prescription medicine that is used with other medicines (such as glucocorticoids) to treat adults with severe
Read moreApproved Use: TAVNEOS® is a prescription medicine that is used with other medicines (such as glucocorticoids) to treat adults with severe active anti-neutrophil cytoplasmic autoantibody (ANCA)-associated vasculitis (granulomatosis with polyangiitis [GPA], formerly known as Wegener’s granulomatosis, and microscopic polyangiitis [MPA]). It is not known if TAVNEOS is safe and effective in children under the age of 18.
Read less